What Is Anatomical Waste?
Anatomical waste is one of a handful of medical waste streams that must be identified and properly segregated prior to storage and disposal in healthcare scenarios. While it might appear that the question, “what is anatomical waste?” is obvious, the answer isn’t always so straightforward. While the Canadian federal government supplies a definition of anatomical waste, so too does every province and territory in the country, all of which may vary slightly in language, terminology, or meaning.
It should also be noted that terminology used by one province can be slightly different than that used by another. For example, anatomical waste is considered to be a subset of biomedical or infectious waste, but in certain scenarios it can be elevated to the level of hazardous waste, which is itself a separate waste category.
Following standard operating procedures as well as guidelines provided by regulatory authorities is essential in properly handling and disposing of anatomical waste.
TOPICS WE WILL COVER:
1 / How does the government define anatomical waste?
2 / Colour-coding and storage of anatomical waste
4 / Daniels Health Canada supports compliance
How does the government define anatomical waste?
While broad guidelines are established by the Canadian Council of Ministers of the Environment for the management of certain waste streams, including anatomical waste, such guidelines are not enforceable unless they are accepted and adopted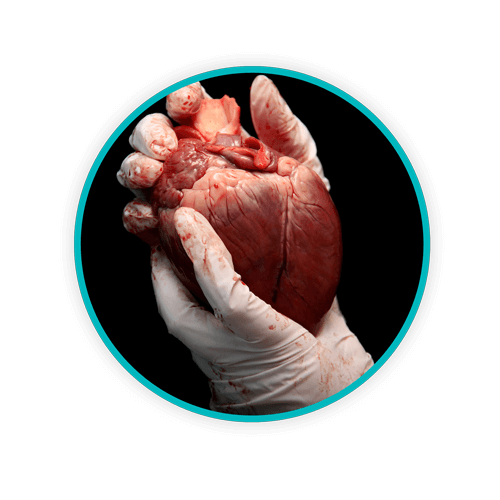 by each province and territory, as well as municipality bylaws.
by each province and territory, as well as municipality bylaws.
For example, the Canadian Biosafety Handbook, provided by the federal government, devotes an entire chapter to waste management. In that chapter, biomedical waste is defined as any kind of waste that is generated in facilities that provide human or animal healthcare services. Such facilities can also include research, training, and clinical testing sites, as well as facilities that produce vaccines.
Biomedical waste must be segregated from general waste streams because it needs to be decontaminated before its disposal. Specific treatment methods are dependent on the standards that are set by each province or territory.
In the aforementioned handbook, human anatomical waste is defined as “all human tissues, organs, and body parts, excluding hair, nails, and teeth.” Another subcategory of biomedical waste is human blood and body fluid waste, although it is not considered to be a biomedical waste after it has been properly decontaminated. By contrast, standard human anatomical waste is still considered biomedical waste even if it’s gone through disinfection or decontamination, potentially requiring specialized disposal based on the province, territory, or locale.
Finding, reviewing, and understanding definitions and processes not only for the federal government, but also for provincial or territorial governments is the first step toward compliant medical waste disposal processes.
Colour-coding and storage of anatomical waste
Containment, labelling, storage, and disposal processes for human anatomical waste can differ slightly between provinces, although federal guidelines must always be followed.
The requirements of provinces and territories throughout Canada follow the guidelines of the federal government in regard to the type of containers, labelling, and storage conditions prior to disposal. Even so, actual disposal and destruction of anatomical waste processes are dependent upon provincial and territorial governments and municipalities. In the Northwest Territories, colour-coding and labelling instructions are specific. Human anatomical waste is to be put into red-coloured bags or containers, while animal wastes are to be placed in orange-coloured bags or containers. 
Biomedical waste management in the province of Ontario also defines biomedical waste as including human anatomical and blood waste, and the same is applicable to animals. Human and animal anatomical waste is to be placed into single-use containers and labelled with the anatomical symbol on a red label.
In the province of Québec, regulations closely mimic those of the Environmental Quality Act in regard to biomedical waste, and states that anatomical biomedical waste is to be treated through incineration. Following incineration, “ash from the incineration of biomedical waste shall be put into rigid, sealed, leak-proof containers after being extinguished and cooled.” Specific guidelines are also stipulated for removing biomedical waste from the site where it was generated, using rigid, sealed, and leak-proof containers, but also requiring that “biomedical waste, other than sharp medical objects or sharp household objects, shall be kept refrigerated at less than 4°C.”
In Saskatchewan, biomedical waste management guidelines are developed by the Provincial Ministry of Health in accordance with various governmental ministries including the Saskatchewan Association of Health Organizations. This province defines human anatomical waste in a similar manner as other provinces under the umbrella of biomedical waste. In Saskatchewan, a number of methods are allowed for the disinfection of biomedical waste, including incineration, autoclaving, microwave treatment, or “any other process that provides disinfection to the required level,” according to the published Saskatchewan Biomedical Waste Guidlines.
A word about storage
As with other aspects of medical waste management processes, requirements for storage of certain waste streams must follow federal guidelines, although every province or territory, as well as municipality within those provinces or territories, may also stipulate their own guidelines, many of which are more stringent.
In Saskatchewan, biomedical waste is to be held in storage areas that identify the area as containing biomedical waste. As such, a placard with the biohazard symbol must be clearly displayed. That waste is to be kept at a temperature of 4°C or lower for any of the materials that are stored for more than four days.
It is the responsibility of every medical care provider in hospitals, care centres, or any other facility that provides medical services to be knowledgeable regarding municipal and provincial guidelines for the management of anatomical waste segregation, containment, storage, and disposal, in addition to the guidelines provided by the federal government.
Daniels Health Canada supports compliance 
Daniels Health Canada offers medical and healthcare facilities access to resources, products, and services to ensure compliance when it comes to healthcare waste disposal processes. For guidance on federal, provincial, or territorial regulations for anatomical waste and other waste streams, and how to improve processes that help to ensure compliance, contact us today.
Let's Talk!
Your time is valuable, and we don’t want to play hard to get. You can either phone us directly on the details listed on our contact page, or feel free to fill out this short form and one of our team members will get back to you as quickly as possible.
